Contents
Improving the Methods of Managing Files and Folders on Your Local Hard Disk Drive
Tagstore is a research software tool developed by the Institute for Software Technology of Graz University of Technology. Its main purpose is to enhance file and folder management on local hard disk drives. Unlike commercial products, Tagstore is made publicly available under the open-source license GPL v3.
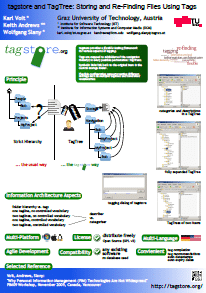
Basic Idea
Tagstore introduces a new approach to filing data by allowing users to tag files and folders instead of organizing them in a traditional folder hierarchy. By doing so, Tagstore generates automatic navigation hierarchies, known as TagTrees, enabling users to navigate through their data using preferred tools such as file browsers or “file open…/” dialogs.
Key Advantages
- Effortless Storage: Users no longer need to browse or select specific folders deep within their directory structures. They can simply choose relevant tags associated with their files during the storage process.
- Intuitive Navigation: Tagstore allows users to navigate to files and folders intuitively through associations rather than remembering exact browsing paths. Tags appear as folders within the TagTrees, making searches more precise as users delve deeper into the structure.
- No Dead Links: Tagstore ensures that all links are active and point to the correct data, eliminating broken links and enhancing usability.
- Transparent Structure: Tagstore does not rely on complex databases, ensuring transparency and confidence in the safety of stored data.
- Smart Tag Recommendations: To simplify the tagging process, Tagstore provides automatic tag suggestions based on file content. Additionally, previously used tags can be quickly accessed by typing a few initial characters.
- ISO Datestamp Tags: Tagstore supports easy organization of data based on processing dates by offering suggested tags in ISO format (YYYY-MM-DD).
- Expiry Dates: Users can set optional expiration dates for stored files, which will then be automatically moved to designated trash folders, promoting organized data management.
- Broad Compatibility: Tagstore works seamlessly across various platforms, including Microsoft Windows, Mac OS X, and GNU/Linux, without requiring users to replace existing software solutions.
- Future Potential: As Tagstore demonstrates its effectiveness as an alternative to traditional folder hierarchies, developers aim to incorporate its features into future operating systems and file systems, providing enhanced user experiences for managing files and folders.
By adopting Tagstore’s innovative approach, users can enjoy a better way of handling files and folders while taking advantage of its numerous benefits.
FAQ
What is tagstore and when would I need it?
Our website provides more information on this topic.
Does tagstore do something with my computer that I don’t want it to?
There’s no need to worry, as tagstore only concerns itself with the items stored within its own directories. Apart from these directories, tagstore only modifies its own configuration files and nothing else.
How much performance does tagstore require?
While there is a background process that monitors the directories for changes, it does not cause significant strain on the computer.
How much storage space does tagstore take up?
Aside from the data you store in tagstore, the program uses less than five megabytes of storage space. However, necessary dependencies (Python, PyWin, PyQt, etc.) can use up to 200 megabytes of space, depending on what is installed and already present on your hard drive.
What does tagstore cost?
Nothing! The research software tagstore is developed under a free license and will be available for free download. The complete source code will likely be published on GitHub in 2012.
Where can tagstore be particularly useful?
Tagstore is generally versatile and not limited to specific data. However, it excels especially as a replacement for folders typically named “Various,” “Miscellaneous,” or “Misc.” Users also have good experiences using tagstore as their default directory for internet downloads.
Can I still use my file browser?
One unique feature of tagstore is that users are not restricted to a specific navigation software. Therefore, you can continue to use your preferred file browser. Also, check out the compatibility section on our website for more information about tagstore.
Can I still use my desktop search engine?
Yes, of course! It is recommended to exclude the navigation directories (not the storage directory) from indexing to prevent duplicate mentions of your files in search results.
Can I still use my backup software?
Yes, of course!
In which languages is tagstore available?
Tagstore is fundamentally multilingual and offered in English and German. If you are interested in another translation, let us know!
Do I have to store all files in tagstore?
No. You can decide for yourself which data should remain in your existing folder hierarchy and which data you want to use with tagstore’s advanced features.
Does tagstore offer any additional comfort functions?
We, as avid users of tagstore, are committed to making tagstore as user-friendly as possible. As such, we have integrated several comfort functions to make working with files easier.
You can automatically assign timestamps to data or have unnecessary files sorted out automatically with defined expiration dates. Read the tagstore details to learn more about its advantages.
Can I store folders in tagstore?
You can store both files and folders in a tagstore. If you create a folder in the storage folder, you will be asked for keywords related to that folder. You can then store any number of files within this new folder, but they will not be keyworded by tagstore.
What advantage does tagstore offer over desktop search engines?
Desktop search engines like Copernic Desktop Search or Google Desktop Search are not competitors to tagstore. In fact, they complement each other well. However, scientific studies show that users generally prefer navigation over searching. Therefore, it is essential that tagstore provides an alternative way of navigating within strict folder hierarchies.
Is there support available for tagstore?
Tagstore is not backed by a company or personal or financial resources. We can only ensure the smooth functioning of a tagstore installation during our scientific test runs. However, if you have programming knowledge, you’ll be glad to know that tagstore has been published under an open-source license. So, you can find bugs independently in the code. Of course, we would appreciate it if bug reports and improvements could find their way back to us, so we can continuously improve tagstore. Please contact us if you find anything. Thank you!
What is the current status of tagstore?
The research program ended in 2012, and we unfortunately cannot continue developing the tagstore research platform further. However, you are welcome to test and use the software because it is free software. Additionally, anyone with Python skills can further develop the software.
Is there a database behind it?
No. The software is straightforward and doesn’t require an unnecessary complicated database. All information is stored in simple text files.
How do I get my data out of tagstore?
Your data in the storage folder remains untouched except for the use of expiration dates and final deletion of a complete tagstore. So, you can move any file out of the storage folder anytime.
The keywords associated with your files are stored in a text file. With at least basic programming knowledge, we can help you access this data.
Can I run multiple tagstores simultaneously?
Yes, as a software, tagstore allows you to operate multiple tagstores concurrently. However, while participating in an official test run, please refrain from using this feature. Afterward, you can certainly create additional tagstores.
This feature is useful for completely separate areas such as photos/others, private/business, ProjectA/ProjectB.
Could you integrate the following function into tagstore?
We welcome suggestions. However, we cannot guarantee that we can provide capacities to implement all wishes.
Does tagstore work with multiple users?
Due to capacity, technical, and organizational reasons, we currently focus on a single user with a local hard drive. Multiuser operation will be part of future research projects.
Is there a synchronization solution for multiple computers?
Due to capacity, technical, and organizational reasons, we currently focus on a single user with a local hard drive. Functions supporting synchronization across multiple computers are technically challenging with tagstore. If you achieve experimental results here, we would love to hear your feedback!
Up to how many files is tagstore effective?
Technically speaking, tagstore implementation works wonderfully up to several thousand files. However, various technical limitations come into play, preventing arbitrary scaling beyond that point.
For specifics, feel free to contact us.
Why is the number of keywords per file limited?
One of the main criteria when creating tagstore was maintaining transparency regarding what happens to your data. That’s why tagstore uses no database. The required links increase exponentially with the number of keywords used. Hence, we must limit the number of keywords per file.
Will tagstore ever become a commercial product?
No. Tagstore is a platform for scientific investigations, is licensed under an open-source license, and will never become a commercial product.
As a student, can I write a paper on the tagstore project?
I’m sorry, but the research project surrounding tagstore ended in 2012. The results are available on the whitepaper page.
Installation
The creation of a new store is not working.
Could it be that you have mistakenly not followed the installation instructions and installed tagstore in the Windows suggested standard folder “C:\Program Files”?
If so, please completely remove tagstore and reinstall it outside of “C:\Program Files”.
After adding a file, nothing happens.
Could it be that you have mistakenly not followed the installation instructions and installed tagstore in the Windows suggested standard folder “C:\Program Files”?
If so, please completely remove tagstore and reinstall it outside of “C:\Program Files”.
Nothing seems to be working.
It may be that you have Python 2.6 or 3.x installed on your system instead of the used version 2.7.
Usage
Why do multiple windows for tagging appear when I add files to my store?
You probably started tagstore several times. Close the process „pythonw.exe“ in the task manager (press Ctrl-Shift-ESC simultaneously) until it only appears once.
Alternatively, you can also restart your system without manually starting „tagstore“.
How can one select tagstore’s suggested tags using the keyboard?
While typing a tag, tagstore suggests tags that begin with the same character sequence. You can easily choose from these suggestions with the mouse by clicking on them. The same result is achieved on the keyboard by selecting the desired tag with the arrow keys and then confirming with the enter key (Enter/Return).
What are those suggested tags next to the input field?
On the left side there are suggestions for tags that can be selected with the mouse. These tags are suggestions based on frequently or most recently used tags as well as an intelligent system that generates suggestions from the file name, user history with similar files etc.
Can all listed files in the tag window be tagged at once with the specified tags?
Of course! Simply select multiple entries as usual (by holding down the control key) and apply the tags as usual.
Additionally, tagstore remembers the entered tags of a tagging process and proposes these as default values for subsequent files. This way, you can tag the first file normally and quickly assign the following files by repeatedly clicking on „Tag“ (or Enter). With this method, you can easily assign identical tags while still having the option to change the selection.
How can already tagged files be retagged or supplemented with additional tags?
Retagging is currently possible in the tagstore manager.
The tagstore manager can be launched either explicitly from the start menu or by choosing the manager button in the tagstore dialog.
How can I delete or rename all links to a file stored in tagstore if necessary?
As soon as a file in the „storage“ folder is deleted or renamed in the „storage“ folder, all corresponding links in the TagTrees are automatically deleted or adjusted accordingly. Therefore, manual editing of a link in the TagTrees is never required.
How can I rename an existing tag for all files?
Renaming a tag is currently possible in the tagstore manager.
The tagstore manager can be launched either explicitly via the start menu or by choosing the manager button in the tagstore dialog.
Is it not possible to simply file away files in the navigation hierarchies instead of tagging them?
Unfortunately, due to technical reasons, filing in the navigation hierarchies could not be implemented as an alternative to tagging. The navigation hierarchies are therefore suitable for accessing files but not for adding new ones.
How can I terminate the background process of tagstore?
There is a tool in the Windows start menu to restart tagstore. To end the background process, open the task manager (Ctrl-Shift-ESC), locate the appropriate pythonw.exe process and end it.
In OS X and Linux, tagstore can be terminated by killing the corresponding Python process. We also provide a start/stop script to start, stop or restart tagstore.

